
by teamneuro-kinesis | Sep 1, 2009 | TImeline
The CGCI platform was granted UL Certification thereby approving the device for use in the medical field. This certification certifies that the CGCI Platform meets all the stringent electrical safety standards required for both electrical safety concerns as well as...

by teamneuro-kinesis | Dec 31, 2008 | TImeline
Additional breakthroughs in the advancement of the CGCI technology platform result in nine patents being submitted relating to the apparatus and method for controlling catheter positioning and orientation, the magnetic linear actuator for deployable catheter tools,...

by teamneuro-kinesis | Dec 11, 2007 | TImeline
An additional nine patients over the course of the preceding year relating to CGCI’s apparatus, method, and system for radar-assistance as well as the first patents for what would become the Optical Catheter are filed. This patent represented the first application for...
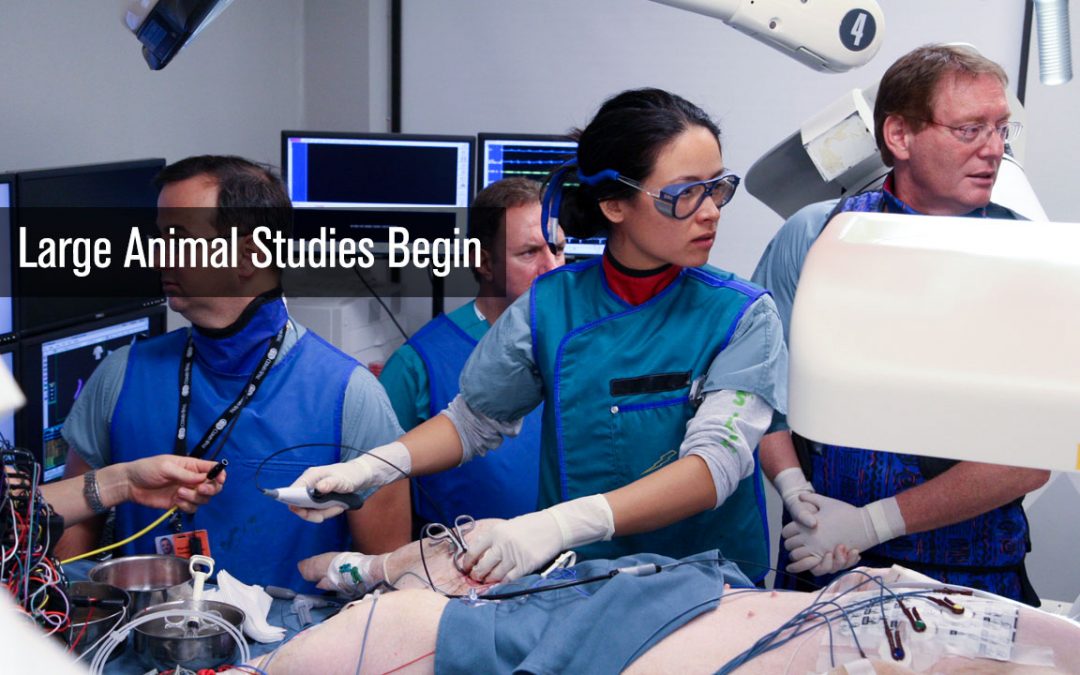
by teamneuro-kinesis | Nov 11, 2007 | TImeline
The first large animal study utilizing the CGCI-1 begins at Cedars Sinai Hospital in Los Angeles, California. The study consisted of 15 subjects undergoing a simulated AFib procedure to show the ability of CGCI to move a catheter to an identified target site, remember...

by teamneuro-kinesis | May 11, 2007 | TImeline
The first fully operational prototype of CGCI is constructed. Built at a 2/3 scale, CGCI-1 featured an 8-lobe model using 18” coils and was capable of generating a magnetic field of 900 Gauss. The unit was installed at Cedars Sinai Hospital in Los Angeles, CA, and was...






